You’ve probably heard of carbon dioxide, the greenhouse gas that’s causing all the fuss these days. In this article, we’ll explore the discovery, properties, applications, and effects of carbon 4 oxide. You’ll learn how this unassuming molecule powered the industrial revolution and how it’s still used today in products you interact with every day.
What Is Carbon 4 Oxide? Carbon dioxide (CO2)
Carbon dioxide (CO2) is a colorless, odorless gas, natural occurring gas, that is present in the atmosphere. CO2 is essential for plant life and is nontoxic in small concentrations, but high levels of CO2 can be dangerous to humans and the environment.
Carbon dioxide is a compound consisting of two oxygen atoms and one carbon atom (CO2). It is produced by the burning of carbon-containing compounds, fermentation, and respiration. Plants take in CO2 during photosynthesis and convert it into glucose and oxygen. CO2 is essential for plant life and growth.
Why is CO2 Called Carbon 4 Oxide?
The numeral IV denotes carbon to represent the oxidation level, not the amount of oxygen. It has to do with electron configuration. In an actual lab, no one refers to CO2 as carbon (IV) oxide, but just carbon dioxide.
The Chemical Structure and Formula of Carbon (IV) Oxide
Carbon dioxide has the chemical formula CO2. This means each molecule contains one carbon atom and two oxygen atoms. The carbon atom is central, with the two oxygen atoms attached, one on each side.
Chemical Structure
Carbon dioxide is a linear molecule, meaning the atoms are aligned in a straight line. The bond angle in a carbon dioxide molecule is 180 degrees. The covalent bond produced in carbon dioxide involves eight electrons shared by the oxygen atoms with the core element, carbon. Carbon is tetravalent, which means it can create four bonds, whereas oxygen can form two. There are four bonds formed, each consisting of two electrons. It follows the octet rule, which requires eight electrons in the valence shell of each atom that each atom’s valence shell. Each oxygen atom has two non-bonding electron pairs.
The linear shape of CO2 means that the molecule has no electric charge. The shared electrons are pulled equally toward the carbon and oxygen nuclei. However, the double bonds between the atoms are quite polar, giving the molecule a large dipole moment. Even though the molecule itself has no charge, it can have strong van der Waals forces, allowing CO2 molecules to form dense gases and liquids.
In its solid and gaseous forms, CO2 molecules are arranged randomly. But when pressurized into a liquid, the molecules align and form a geometric crystal structure. The density of liquid and solid CO2 are higher than the gaseous form because the molecules are packed more closely together.
At standard temperature and pressure, carbon dioxide exists as a colorless, odorless gas. It has a linear geometry with octet stability around the carbon atom. The double bonds between carbon and oxygen are very polar, although the symmetrical shape of CO2 gives it no net charge or polarity. This unique set of properties allows carbon dioxide to play an important role in many areas of science and industry.
Knowing the chemical formula and structure of carbon dioxide will help you understand its key characteristics and behavior.
Physical and Chemical Properties of Carbon Dioxide
Carbon dioxide (CO2) is a colorless, odorless gas composed of one carbon and two oxygen atoms. It has some important properties:
Density
At standard temperature and pressure, CO2 is denser than air, with a density of 1.98 kg/m3, about 1.53 times as heavy as air. Its density is 1.80 g L-1 at 25°C and 1 atm. Carbon dioxide has no liquid state at pressures below 0.51795(10) MPa (5.11177(99) atm). This means it will accumulate in low areas and can cause asphyxiation.
Solubility
CO2 is moderately soluble in water (1.79 volumes per volume at 0 °C and atmospheric pressure, larger amounts at higher pressures), dissolving to form carbonic acid (H2CO3). Its solubility decreases as temperature increases. CO2 dissolved in water is what gives many carbonated beverages their fizz.
Acidity
When CO2 dissolves in water, it forms H2CO3, which then dissociates into hydrogen (H+) and bicarbonate (HCO3−) ions. This gives carbonated water a slightly acidic pH between 3 to 4.
Greenhouse Gas
CO2 is a greenhouse gas, meaning it absorbs and emits radiation within the thermal infrared range. In large amounts, CO2 and other greenhouse gases contribute to the greenhouse effect and global warming.
Non-flammable
Although CO2 contains carbon and oxygen, it will not burn or support combustion. Its high stability makes it useful for fire extinguishers.
Reacts with bases
CO2 reacts with bases like hydroxides (OH−) and amines (NH2) to form salts. With hydroxides, it forms carbonates (CO3 2−); with amines, it forms carbamates (NHCOO−).
Forms carbonates
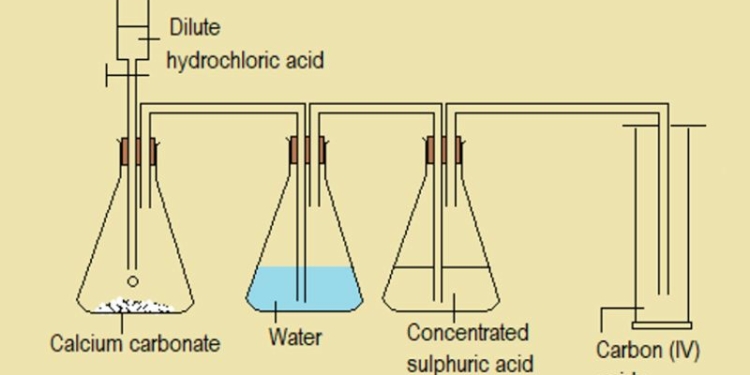
When CO2 reacts with metal oxides, it forms metal carbonates. For example, with calcium oxide (CaO), it forms calcium carbonate (CaCO3). This reaction is used industrially to produce limestone and other carbonates.
The unique properties of carbon dioxide make it essential for many applications like carbonated beverages, fire extinguishers, and the production of fertilizers and limestone.
The Discovery and History of Carbon Dioxide
According to the encyclopedia, Carbon dioxide (CO2) was first discovered in the 17th century by Jan Baptista van Helmont (c. 1580–1644; some sources give the death date as 1635), a Flemish chemist. Around 1630, he produced CO2 by burning wood in a closed vessel and noticed that the remaining gas extinguished flames, at the time thought to be a single substance or element. Van Helmont coined the word “gas” to describe these vapors and collected the gas given off by burning wood, calling it gas sylvestre, which means (“wood gas”).
Today, this gas is known as CO2. Van Helmont is credited with the discovery. He also noticed that wine fermentation and other natural processes produce carbon dioxide. Other scientists soon noticed similarities between the processes of respiration and combustion, both of which consumed and emitted carbon dioxide. For example, they discovered that when a candle flame is trapped in a jar with a restricted supply of oxygen, it eventually goes out.
In 1756, Scottish chemist Joseph Black studied the properties of CO2 further. He found that CO2 was denser than air, that it was present in the atmosphere, and that it combined with other chemicals to form new compounds. Black called the gas “fixed air” because it was chemically combined in carbonate rocks. Black also recognized carbon dioxide in exhaled breath, determined that it is heavier than air, and described its chemical properties as those of a weak acid. Van Helmont and Black’s pioneering work quickly led to the discovery of other gases by English scientist and physicist Henry Cavendish (1731-1810), French chemist Antoine-Laurent Lavoisier (1743-1794), Swedish chemist Carl Wilhelm Scheele (1742-1786), and others.
In the mid-1700s, English chemist Joseph Priestley (1733-1804) developed the first practical application for carbon dioxide. Priestley reproduced Black’s tests with a gas produced by grain fermentation and demonstrated that it had the same qualities as Black’s fixed air or carbon dioxide. When Priestley dissolved the gas in water, he discovered that it produced a delightful beverage with a somewhat acidic flavor. This was the first artificially carbonated beverage, sometimes known as soda water or seltzer. Carbon dioxide is still used to manufacture colas and other soft drinks. In addition to providing bubbles and zest, the gas serves as a preservative.
In the 19th century, scientists began to recognize the importance of CO2 in the environment. Eunice Foote, an American scientist, conducted experiments in 1856, decades before the phrase “greenhouse gas” was invented, Eunice Newton Foote demonstrated the greenhouse effect in her home laboratory. She placed a glass cylinder filled with carbon dioxide in sunlight and discovered that it heated up significantly more than a cylinder of regular air.
John Tyndall, an Irish physicist, found that CO2 and water vapor strongly absorbed heat radiation. These discoveries established the role of CO2 as a greenhouse gas that traps heat in the atmosphere.
In nature, carbon dioxide plays an essential role in maintaining Earth’s temperature balance through the greenhouse effect. However, human activities like burning fossil fuels have caused CO2 levels to increase dramatically, trapping more heat in the lower atmosphere. This human-caused increase in atmospheric CO2 is the main driver of current global climate change, according to the vast majority of climate scientists.
Uses of Carbon 4 Oxide?
Carbon dioxide, or CO2, has several important uses in various industries. Here are some major examples:
Carbonated Beverages
Carbon dioxide is used to carbonate beverages like soda, beer, and sparkling water. The CO2 is dissolved into the liquid under pressure and then released when you open the can or bottle, creating a bubbly fizz. Without CO2, popular sodas like Coca-Cola and Pepsi wouldn’t have their signature bubbles.
Fire Extinguishers
CO2 is commonly used in fire extinguishers because it helps smother the fire by displacing oxygen. The carbon dioxide is stored as a compressed gas and then released when the extinguisher is triggered. It works for various types of fires but works best on electrical or grease fires. The carbon dioxide smothers the fire by depriving it of oxygen.
Preservation and packaging
Carbon dioxide is used to preserve and protect perishable foods like meat, fish, and produce. When foods are packaged in a CO2 environment, it slows down spoilage and oxidation, allowing foods to stay fresh longer. The CO2 displaces oxygen, inhibiting the growth of bacteria and microbes.
Agriculture
Carbon dioxide is essential for plant life and growth. It is commonly used in greenhouses to help plants thrive, especially during the winter months. The increased CO2 concentration speeds up photosynthesis, allowing plants to grow faster and produce higher yields. Many farmers use CO2 generators or burners to enrich greenhouse air with carbon dioxide.
Welding and cutting
Carbon dioxide is used as a shielding gas in welding applications like gas welding, laser welding, and plasma welding. An inert gas like CO2 protects the weld from oxidation and contamination. The carbon dioxide creates an oxygen-free environment around the weld as the metal is heated and fused. This results in a cleaner, higher-quality weld. CO2 is also used as a cutting gas to cut through metals in oxyfuel-cutting processes. It is also used in laser cutting to help blow away debris and slag from the cutting area.
pH Control and Solvent
Carbon dioxide is used to regulate the pH in swimming pools and spas. When the pH rises too high, CO2 can be bubbled into the water to lower it to the proper range. The carbon dioxide produces carbonic acid, which lowers the alkalinity. Proper pH regulation is important for safe, balanced pool chemistry. Carbon dioxide is also used in some industrial applications as a pH adjuster. It is also used as a solvent in some dry cleaning and processing applications.
Oil Recovery
In the petroleum industry, carbon dioxide is used for enhanced oil recovery. The CO2 is injected into oil reservoirs to help recover more oil that would not otherwise flow out on its own. The carbon dioxide helps dissolve and separate the oil from the rock so it can flow more freely. This helps extend the productivity of oil fields.
Other uses of carbon IV Oxide include the following:
- In the medical field, CO2 is used for insufflation to expand body cavities during surgery and to stimulate breathing in critical care patients.
- It is commonly used as a refrigerant in refrigerators and air conditioners because of its ability to absorb and release heat.
- CO2 is used in the production of dry ice, which is frozen CO2. Dry ice is used to preserve perishable goods during transport and to create special effects like fog.
Effects of Carbon Dioxide
Carbon dioxide (CO2) has several concerning effects on our environment. Here are seven of the major impacts:
Climate change
The increased CO2 in the atmosphere acts as a greenhouse gas, trapping heat in the lower atmosphere. This results in rising global temperatures, which leads to rising sea levels, melting glaciers, more extreme weather events, worsening wildfires, etc. The impacts of climate change are already apparent and will only intensify if CO2 levels continue to rise.
Ocean acidification
When CO2 is absorbed into the oceans, it reacts with seawater to form carbonic acid. This makes the water more acidic, which affects marine life like coral, plankton, and shellfish. Their shells and skeletons are made of calcium carbonate, which dissolves in acidic conditions. This threatens entire food chains and ecosystems.
Health issues
Exposure to high levels of CO2 can cause a range of health problems in humans, like respiratory issues, headaches, dizziness, and impaired cognitive function. In poorly ventilated indoor spaces, CO2 buildup is a concern. CO2 isn’t directly toxic, but high levels indicate poor air circulation and ventilation.
Plant growth
While CO2 is essential for photosynthesis, excess amounts can also be detrimental to plant life. Some plants may experience initial increased growth and yield, but long-term effects include reduced protein content, increased susceptibility to pests and disease, and changes in flowering times. This could disrupt ecological relationships and food webs.
Extreme weather
Rising CO2 levels are linked to changes in weather patterns that lead to more frequent or intense extreme weather events like heat waves, droughts, heavy rain and snowfall events, and hurricanes. These extreme weather events pose risks to agriculture, infrastructure, and public health.
Sea level rise
As the climate continues to warm due to excess CO2, sea levels are rising at an accelerating rate due to the thermal expansion of water and melting land ice like glaciers and ice sheets. Sea level rise threatens coastal communities and islands, shoreline ecosystems, and freshwater resources. Low-lying areas are the most vulnerable.
Melting permafrost
Vast amounts of carbon and methane are stored in Arctic permafrost. As the climate warms, this permafrost is thawing and releasing additional greenhouse gases, creating a feedback loop. The impacts include:
- Damage to infrastructure.
- Changes to landscapes and habitats.
- The potential for trapped methane to further accelerate global warming.
Reducing carbon IV oxide emissions and stabilizing climate change will require a concerted global effort to transition from fossil fuels to renewable energy and make societies more sustainable and eco-friendly overall. Although carbon dioxide has been crucial for life on Earth and modern civilization, maintaining a balanced carbon cycle is key to the long-term health of our planet.
What is the Difference Between Carbon 2 Oxide and Carbon 4 Oxide?
Carbon dioxide (CO2) and carbon monoxide (CO) are two very different compounds, even though their names seem quite similar. C02 contains one carbon atom and two oxygen atoms, giving it the chemical formula CO2. Carbon monoxide, on the other hand, has one carbon atom and one oxygen atom, so its formula is CO.
Carbon dioxide is a colorless, odorless gas that is naturally occurring and essential for life on Earth. Plants take in carbon dioxide and use it for photosynthesis, releasing oxygen as a byproduct. Too much CO2 in the atmosphere, however, contributes to global warming and climate change. Carbon monoxide, conversely, is a highly toxic and colorless, odorless gas produced by the incomplete combustion of carbon-containing compounds like gasoline.
In terms of their chemical structures, carbon dioxide has a linear geometry with two double bonds between the carbon and oxygen atoms. Carbon monoxide has a simpler linear structure with a triple bond between the carbon and oxygen. The properties of compounds depend strongly on their geometries and bonds, so CO2 and CO behave very differently. Carbon monoxide has no major commercial uses due to its toxicity. It is primarily produced as a byproduct of combustion and must be carefully monitored and vented.
In summary, while carbon dioxide and carbon monoxide may sound quite similar, they have very different chemical properties, structures, and uses. One is essential for life, while the other is toxic. It’s important to understand the difference between these two colorless, odorless gases.
Frequently Asked Questions about Carbon 4 Oxide
Here are some of the most frequently asked questions about Carbon dioxide, or CO2, and their answers.
Where does carbon dioxide come from?
CO2 is produced naturally through respiration, volcanic eruptions, and the decay of organic matter. However, human activities like burning coal, oil, and gas for energy and transportation have caused CO2 levels to increase by over 40% since the Industrial Revolution. Deforestation also contributes to higher CO2 because trees absorb CO2 from the atmosphere. When forests are burned or decay, the CO2 is released back into the air.
How does carbon dioxide affect the climate?
In normal amounts, CO2 helps keep the planet’s temperature regulated through the greenhouse effect. However, excess CO2 enhances this effect by trapping more heat in the lower atmosphere. According to the EPA, CO2 levels today are the highest they’ve been in at least 800,000 years. The increased heat is causing rising sea levels, melting Arctic sea ice, more extreme weather events, worsening wildfires, and other severe effects that constitute climate change. Reducing CO2 emissions is critical to mitigating and avoiding the most catastrophic consequences of climate change.
How can we reduce carbon dioxide emissions?
To curb rising CO2 levels and limit climate change, we need to make big reductions in greenhouse gas emissions. This includes transitioning from fossil fuels to renewable energy like solar and wind, driving fuel-efficient vehicles, improving energy efficiency, and conserving natural habitats like forests and wetlands. Individual actions like reducing waste and eating less meat can also help lower your carbon footprint. Together, these steps can make a difference in creating a more sustainable future for our planet.
Conclusion
Carbon dioxide, or CO2, is a greenhouse gas that is naturally occurring and harmless in small amounts; however, when levels rise, it can have an impact on productivity and sleep. CO2 levels are highest inside with less ventilation, as they are mostly produced by the air we exhale. As you have seen, there is more to carbon 4 oxide than meets the eye.








Discussion about this post